The FDA recently approved Novartis' Cosentyx (secukinumab) for
the treatment of non-radiographic axial spondyloarthritis, a form of axial
spondyloarthritis (axSpA), in adult patients. Below is a look at market access
for ankylosing spondylitis, the more severe form of axSpA in which inflammatory
arthritis of the spine is visible on X-ray. For that indication, most
medications are covered under both the pharmacy and medical benefit with some
limitations. For all of the drugs, more than half of covered lives are under
the preferred tier/preferred with prior authorization or step therapy and
covered tier/covered with PA/ST under the pharmacy benefit.
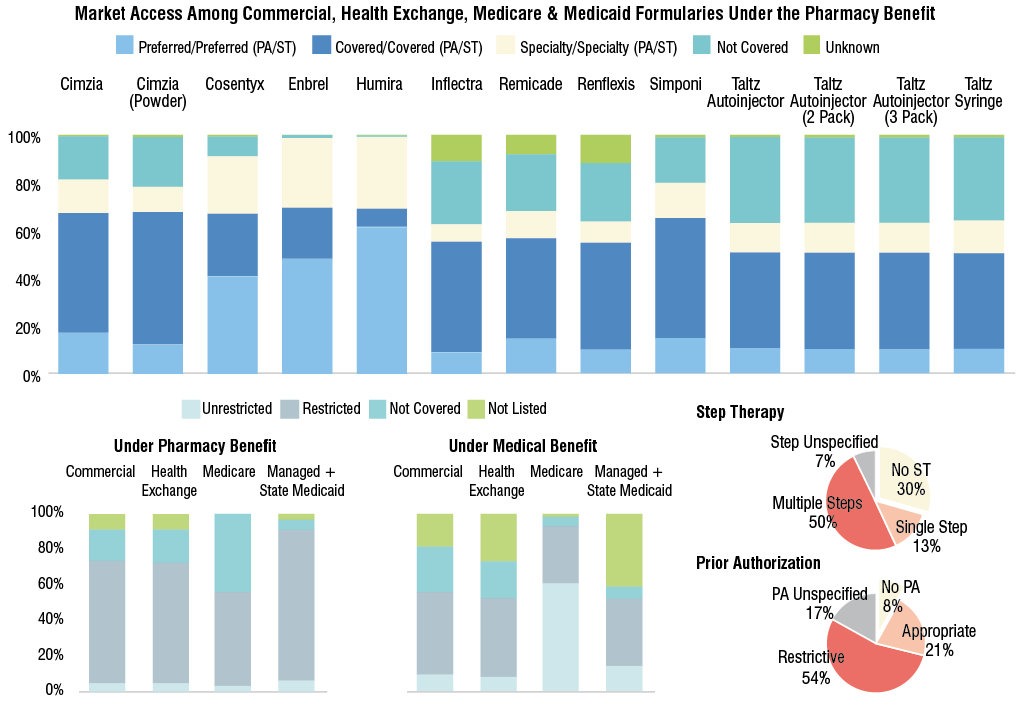
NOTES: Under the pharmacy benefit, the numbers of total covered
lives under commercial, health exchange, Medicare and Medicaid formularies are
178.3 million, 10.0 million, 46.4 million and 63.1 million, respectively. Under
the medical benefit, the numbers of total covered lives under commercial,
health exchange, Medicare and Medicaid formularies are 176.2 million, 10.0
million, 48.4 million and 63.1 million, respectively.
SOURCE: Managed Markets Insight & Technology, LLC database as of June 2020.
SOURCE: Managed Markets Insight & Technology, LLC database as of June 2020.
I'm glad I found this web site, I couldn't find any knowledge on this matter prior to.Also operate a site and if you are ever interested in doing some visitor writing for me if possible feel free to let me know, im always look for people to check out my web site. mao inhibitor guide
ReplyDelete